Down the Drain:
Energy Consumption at the Berkeley YMCA Pool
A Vital Signs Case Study
Prof. Alison Kwok/ARCH 507/University of Oregon Architecture Dept.
Data Analysis
Team Members:
Anne Deutsch
Ram Ganapathy
Geoff Grummon
Data Analysis
Determining the Total Heat Loss from the Pool
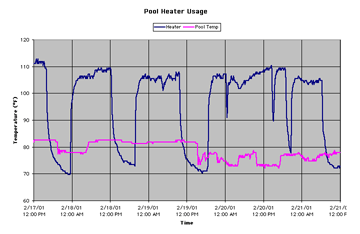
The amount of energy consumed by the pool heater was determined by measuring how long the heater was on over a given time period. The energy that the heater put into the pool is equal to the total amount of heat loss, because the water temperature is maintained at a constant 82 °F. The graph shows the heater cycles over a four-day period. When the heater case was around 110 °F, the heater was running, and when the case was at room temperature the heater was off.
Note that the line indicating pool temperature drops off sharply in some places. Because the pool could never cool off this quickly, we suspect that the HOBO thermistor was splashed out of the water or disturbed.
As you can see, the pool heater ran regularly and for extended periods of time. Over 4 days, it ran 64 hours and 30 minutes, or about 16.125 hours a day. From the heater specifications, we found that it uses about 500,000 BTU per hour.
16.125 hrs running/day
@500,000 BTU/hr
= 8,062,500 BTU used per day
Since this heater runs at about 75% efficiency, we estimated that 6,046,875 BTU/day go into the pool.
The current price of natural gas in California is approximately $1.28 per therm (one therm equals 100,000 BTU/hr). Assuming that gas prices stay similar to this, and that pool heating patterns remain the same, the yearly energy cost can be calculated as follows:
=8,062,500 BTU used per day
@ $1.28 per 100,000 BTU/hr
= $103.20 per day
= $37,668 per year
Heat Loss from Evaporation
The energy loss from evaporation was calculated using an ASHRAE equation:
Wp = A(Pw-Pa) (95+0.425V)/Y
where
Wp = the rate of evaporation (lb/hr)
A=pool area in (sq. ft.)
Pw = sat. vapor pressure at surface water temp (in. Hg)
Pa = sat. pressure at room air dew point (in. Hg)
V = air velocity over water (fpm)
Y = latent heat req. to change water to vapor at water temp (btu/lb)
Using this equation and the data collected in the pool room, the rate of evaporation was estimated to be 194.3 pounds per hour (about 24 gallons per hour) or 4663.2 pounds per day. Knowing that water near 80 °F contains about 1,047 BTU/lb, we could determine the amount of heat loss through evaporation.
4663.2 lb/day
@ 1,047 BTU/lb
= 4,882,370 BTU lost per day through evaporation
We also measured evaporation by measuring the amount of water in a tub in the pool room over a period of two hours. The amount of evaporation found by this method was twice the amount determined above. This number cannot be considered accurate, because our measuring instruments were not reliable, and the amount of heat loss would be greater than the amount of heat input from the heater.